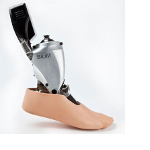
Cogmedix announced that it is a named supplier to BiOM Ankle, the Silver Winner in the rehabilitation and assistive-technology product category of the 2012 Medical Design Excellence Awards (MDEA) competition. The 2012 MDEA winning products, and their suppliers, were announced at a presentation ceremony held May 23, 2012 inside the Philadelphia Marriott Downtown hote l. The ceremony was held in conjunction with the MD&M East Event at the adjacent Pennsylvania Convention Center.
The BiOM Ankle was manufactured and submitted by iWalk Inc., with supply [device contract manufacturing] credit to Cogmedix. Cogmedix manufacturers iWalk’s BiOM product at Cogmedix’s cGMP facility in West Boylston, providing services such as inventory management, assembly, test, final packaging and order fulfillment.
Presented by UBM Canon, and sponsored by Medical Device and Diagnostic Industry magazine, the MDEA program is the MedTech industry’s premier competition for medical device design and innovation. It recognizes the achievements of medical device manufacturers, their suppliers, and the many people behind the scenes — engineers, scientists, designers, and clinicians — who are responsible for the groundbreaking innovations that are changing the face of healthcare. MDEA – winning products excel in the areas of product innovation, design and engineering achievement, end-user benefit, and cost-effectiveness in manufacturing and healthcare delivery.
A comprehensive review of the entries was performed by an impartial, multidisciplinary panel of third-party jurors with expertise in biomedical engineering, clinical practice, diagnostics, human factors, industrial design, manufacturing, and medicine. Entries are evaluated on the basis of their design and engineering features, including innovative use of materials, user-related functions that improve healthcare delivery and change traditional medical attitudes or practices, features that provide enhanced benefits to the patient, and the ability of th e product development team to overcome design and engineering challenges so that the product meets its clinical objectives. The 2012 MDEA Jury selected 25 winning products out of 41 finalists in 10 medical product categories.
“With iWalk’s selection of Cogmedix as its core medical device contract manufacturing partner, iWalk is able to focus on their core technology development, continued product innovation, and marketing efforts,” said Matt Giza, Vice President and General Manager of Cogmedix. “We have a very collaborative relationship and iWalk can feel confident that the BiOM Ankle is built in an environment that has strict compliance adherence in its DNA.”